

Ready to eat (RTE) meat products can serve as vehicles for a range of zoonotic and environmentally persistent viruses. As consumer demand grows for high‑quality, minimally processed and safe products, High Pressure Processing (HPP) stands out as a robust, sustainable, and validated solution to address both current and emerging viral threats. HPP is a nonthermal technology for reducing microbiological risks in RTE meat and other related categories. This article summarizes current scientific understanding for applying HPP to real world applications to reduce viral risks for and briefly discusses other foodborne viruses of concern.
Ready‑to‑eat (RTE) meat products due to contamination during primary production, the viral presence in animal tissues, and handling steps that occur after lethality treatments. Unlike vegetative bacteria, many viruses demonstrate high stability against desiccation, moderate acidity, salinity, and refrigeration, enabling their survival in minimally processed meat matrices. The meat industry is facing increasing challenges related to viral pathogens in raw tissues, organ‑based products and processed meats. These products could pose a potential environmental risk if an effective inactivation barrier is not applied.
High Pressure Processing (HPP) as a key ally
As the market shifts towards mild‑processed and RTE formulations, the role of nonthermal interventions becomes increasingly critical to mitigate residual viral infectivity across the meat supply chain while preserving delicate sensory attributes. High Pressure Processing (HPP) has gained significant recognition as a reliable technology capable of enhancing food safety while maintaining product quality. As the industry faces increasing challenges linked to viral pathogens such as African Swine Fever virus, Hepatitis E virus and Avian Influenza, the role of HPP becomes more relevant than ever.

Key viruses affecting RTE meat and their response to HPP
African Swine Fever virus (ASFV)
African Swine Fever virus (ASFV) is a large, enveloped DNA recognized as one of the most economically disruptive viruses for the global pork industry, due to its environmental persistence and high mortality rates. Although ASFV is not a zoonotic agent and poses no risk to humans, its detection in animal tissues and pork-derived products remains relevant from a biosecurity perspective, as contaminated raw materials can contribute to indirect transmission within the swine production chain.
Regarding HPP treatment, Petrini, et al. (2024) demonstrated the sensitivity of ASFV to high pressure: applying a pressure of 600 MPa/87,000 psi for 3-7 min on three different pork tissues homogenates (spleen, kidney, and loin). The viral load was reduced by more than 7-log units, even with the shortest holding time.
Although foodborne transmission is not the primary route of concern, ingestion of contaminated pork materials is recognized as a potential indirect pathway for spreading ASFV between animal populations. In this context, HPP can serve as an additional post‑process control step in regions facing active outbreaks or elevated sanitary risk. ASFV detection in pork tissues has major economic implications, as outbreaks lead to immediate trade restrictions, movement bans and large‑scale culling programs. These measures generate substantial financial losses for the swine industry, and recent evidence suggests that validated HPP treatments could support the exemption of certain processed products from trade suspensions, strengthening biosecurity within integrated control strategies.
Hepatitis E virus (HEV)
Hepatitis E (HEV) is increasingly recognized as one of the primary foodborne viral hazards associated with pork‑based RTE products, including pâtés, liver products, and certain minimally heated processed meats. Fat content, water activity and pH strongly influence HEV susceptibility to HPP, which requires case-by-case assessment (Nasheri, et al., 2020; Johne et al., 2021).
In a study focusing on prosciutto, Lee et al. (2023) demonstrated that HPP at 400 MPa/58,015 psi reduced viral RNA copies by approximately 1.6-log units, with no further inactivation at higher pressure levels. However, significant log reductions have been observed in the 600 MPa/87,000 psi for 2-5 min range, depending on the matrix and product formulation: Pellerin et al., (2026) demonstrated the effectiveness of HPP against HEV showing no presence of residual infectious particles after a treatment of 600 MPa/87,000 psi for 1 min in pork liver. In human milk, Bouquet et al., (2023) showed that a HPP treatment of 600 MPa/87,000 psi for 5 min led to a virus inactivation like that obtained after holder pasteurization, a gold standard procedure for inactivating HEV. These findings collectively underscore the potential of HPP as a strategy for mitigating HEV contamination in food products.
Avian Influenza (AI) in meat products
Highly Pathogenic Avian Influenza (HPAI) has caused notable disruption worldwide in recent years, raising questions around food safety despite the extremely minimal risk of foodborne transmission. Research from specialized virology centers indicates that HPAI viruses are sensitive to HPP, showing significant reductions when treated under standard industrial parameters (500-600 MPa/79,800-87,000 psi). Processing at 500 MPa/ 79,800 psi for only 25 seconds inactivated more than 5-log PFU/ml of the H7N7 serotype in both cell culture medium and chicken meat homogenate (Figure 1; Isbarn et al., 2007). Also processing at 600 MPa/87,000 psi for 1 min resulted in more than a 6-log reduction in the virus, in a chicken meat homogenate (Buckow, et al., 2017).
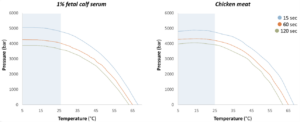
The results of the study suggest that HPP is particularly effective in strengthening the case for its use as part of an enhanced safety strategy for ready-to-eat (RTE) poultry products.
We also discussed avian influenza and other viruses in detail during one of our webinars about Pet Food: HPP’s Role in Raw Pet Food Safety Amid Avian Influenza Concerns.
Other relevant viruses: Hepatitis A (HAV) and Norovirus
Hepatitis A (HAV) and norovirus remain the top global causes of viral foodborne illnesses, affecting a wide range of other RTE categories. Relatively low-pressure levels (250-400 MPa/36,300-58,000 psi) may inactivate HAV in shellfish and fruit and vegetable preparations (Calci et al., 2005; Kingslei et al, 2005). Additionally, the available scientific evidence also suggests that HPP may reduce the risk associated with Norovirus, especially when consuming foods with a higher risk, such as berries and raw oysters. At 550 MPa/79,700 psi for 2 min, Human Norovirus (HuNoV) present in whole strawberries, blueberries, raspberries and its purees were inactivated by >3.0-log virus particles/g (Huang et al., 2016).
Studies on norovirus surrogates suggest that for the consumption of raw oysters, reductions greater than 4-log PFU can be achieved by applying a pressure of 400-600 MPa/58,000-87,000 psi (Li et al., 2009; Li et al., 2013). Furthermore, a study of HuNov observed a 1.9–2.0 log reduction in viral genomic equivalent copies (GEC) after HPP treatment at 400 MPa/58,000 psi for 5 min in naturally contaminated or inoculated oysters Imamura et al. (2017). Another study involving human volunteers reported no cases of infection from HuNov oysters treated at 600 MPa/87,000 psi for 5 min (Leon et al., 2011).
If you are curious about this topic, you can read more about foodborne viral inactivation and HPP on our whitepaper at the following link.











