In the ever-evolving landscape of food safety and consumer preferences, High-Pressure Processing (HPP) emerges as a powerful ally. This cold pasteurization method utilizes intense pressure to extend shelf life while preserving the natural attributes of food. But what if we could enhance its effectiveness even further? Enter clean label antimicrobials, a strategic combination that aligns with consumer demand for minimally processed foods.
High Pressure Processing (HPP), also known as high hydrostatic pressure, is a food preservation technology that aligns with both the food industry needs and emerging consumer trends. HPP involves subjecting packaged food products to intense hydrostatic pressure, which inactivates harmful microorganisms while preserving the nutritional quality and sensory attributes of the products. As consumers increasingly seek minimally processed, clean-label foods, HPP emerges as a nonthermal method that extends shelf life without compromising safety or quality. By ensuring pathogen reduction and maintaining freshness, HPP contributes to a safer and more appealing food supply.
However, HPP is not a sterilization technique. This means that pressure-resistant microorganisms will survive the process, so additional controls are required to prevent their growth through shelf life. In this regard, bacterial spores and sublethal injury on vegetative forms represent major HPP industrial challenges for the stability of food products. Furthermore, slight changes in quality attributes (such as color or texture) may occur when harsher processing conditions are employed to control of pressure-resistant microorganisms. These alterations may harm the product acceptance by the consumer market.
Therefore, HPP treatment at “milder conditions” is feasible alternative assuring quality, safety and economic advantages. This is where clean-label antimicrobial ingredients come into play. These ingredients, such as plant extracts, or natural ferments (like nisin, cultured dextrose, salts derived from organic acids), can complement HPP by providing an additional layer of safety against pathogens. By combining both strategies it is possible to achieve greater effectiveness in food preservation without compromising quality or safety.
The multi-hurdle approach: combining HPP and natural antimicrobials
Combining HPP and clean label antimicrobials create a multi-hurdle system with the following benefits:
- Immediate synergy: Each hurdle—pressure and antimicrobial action—complements the other during treatment. Together, they form a robust defense against spoilage and pathogens.
- Sub-Lethal Injury Control: Some microorganisms survive HPP but remain sub-lethally injured. Clean label antimicrobials step in, preventing their revival during storage.
- Extended Shelf Life: The synergy extends product shelf life beyond what either method achieves alone.
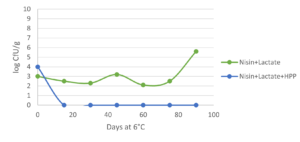
 Innovative applications for hurdle technology by combining natural antimicrobials are well documented. The use of antimicrobials (nisin, lactate and in combination) alone on cooked ham is not able to control Listeria monocytogenes. However, combining them with HPP (600 MPa / 87,000 psi for 5 min) ensure that the pathogen remains below the detection level (<1 CFU/g) even at 6 °C / 42.8 °F during more than 90 days (Jofre et al., 2008; fig. 1). Similarly, the combination of HPP (400 MPa / 58,000 psi for 10 min), enterocins (a clean label bacteriocin) and refrigerated storage (1 °C / 33.8 °F) served to control L. monocytogenes growth (<4 MPN/g) in sliced cooked ham during 84 days, even when the cold chain was broken for 24 h after 60 days of storage (Marcos et al., 2008). This suggests that less intense HPP conditions (400 MPa / 58.000 psi) can be used in combination with natural antimicrobial compounds cand yield equivalent results to those achieved with standard harsh HPP parameters (600 MPa / 87,000 psi).
Innovative applications for hurdle technology by combining natural antimicrobials are well documented. The use of antimicrobials (nisin, lactate and in combination) alone on cooked ham is not able to control Listeria monocytogenes. However, combining them with HPP (600 MPa / 87,000 psi for 5 min) ensure that the pathogen remains below the detection level (<1 CFU/g) even at 6 °C / 42.8 °F during more than 90 days (Jofre et al., 2008; fig. 1). Similarly, the combination of HPP (400 MPa / 58,000 psi for 10 min), enterocins (a clean label bacteriocin) and refrigerated storage (1 °C / 33.8 °F) served to control L. monocytogenes growth (<4 MPN/g) in sliced cooked ham during 84 days, even when the cold chain was broken for 24 h after 60 days of storage (Marcos et al., 2008). This suggests that less intense HPP conditions (400 MPa / 58.000 psi) can be used in combination with natural antimicrobial compounds cand yield equivalent results to those achieved with standard harsh HPP parameters (600 MPa / 87,000 psi).
A
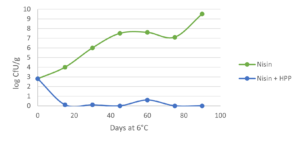
B
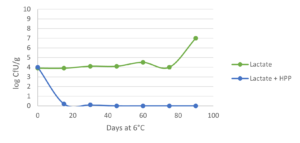
C
There are other examples of food grade antimicrobial compounds that have demonstrated synergism with HPP like: plant-origin antimicrobial agents (essential oils, oleoresins and vegetal extracts), acids from natural fermentation (vinegars) and their salts, and animal/microbial-origin compounds (antimicrobial peptides, active lipids, chitosan, lactoperoxidase systems, lysozyme, bacteriocins), competitive flora and bacteriophages or their endolysins.
In summary, the marriage of HPP and clean label antimicrobials aligns with consumer desires for natural, safe, and minimally processed foods. The major improvements have pointed out that effective microbial inactivation rates may be reached at mild HPP set ups, assuring reduced energy consumption and maintenance costs, and maximizing processing throughput by effective shortened cycles (higher productivity in cycles/per hour).
 Discover the potential of this combo approach at Hiperbaric’s HPP Innovation Week 2024. The event is set to occur from June 18 to June 20, 2024. The session titled ‘Clean-label antimicrobials in synergy with HPP’ on the second day will explore this exciting topic. Don’t miss this chance to gain insights into the cutting-edge applications of HPP in the food industry. Register now for free access to the event.
Discover the potential of this combo approach at Hiperbaric’s HPP Innovation Week 2024. The event is set to occur from June 18 to June 20, 2024. The session titled ‘Clean-label antimicrobials in synergy with HPP’ on the second day will explore this exciting topic. Don’t miss this chance to gain insights into the cutting-edge applications of HPP in the food industry. Register now for free access to the event.











